Medical Technology kicked up a Notch: Novanta
We produce Insufflators, Medical Pumps, Couplers and Tube Sets that simplify and facilitate diagnosis and treatment easier for everyone. Together with customers, doctors, and technical experts, our teams continuously search for new ways to improve patient care.
Quality First. In Everything we do.
At the forefront of minimally invasive surgery, our commitment to uncompromising quality drives every innovation. With rigorous standards, expert craftsmanship, and cutting-edge technology, we empower healthcare professionals to deliver safer, smarter care.
Product Lifecycle
At every stage of the product lifecycle — our teams bring unmatched expertise and unwavering support. We don’t just deliver products; we build lasting partnerships. You can count on us to be by your side from day one — and every day after.
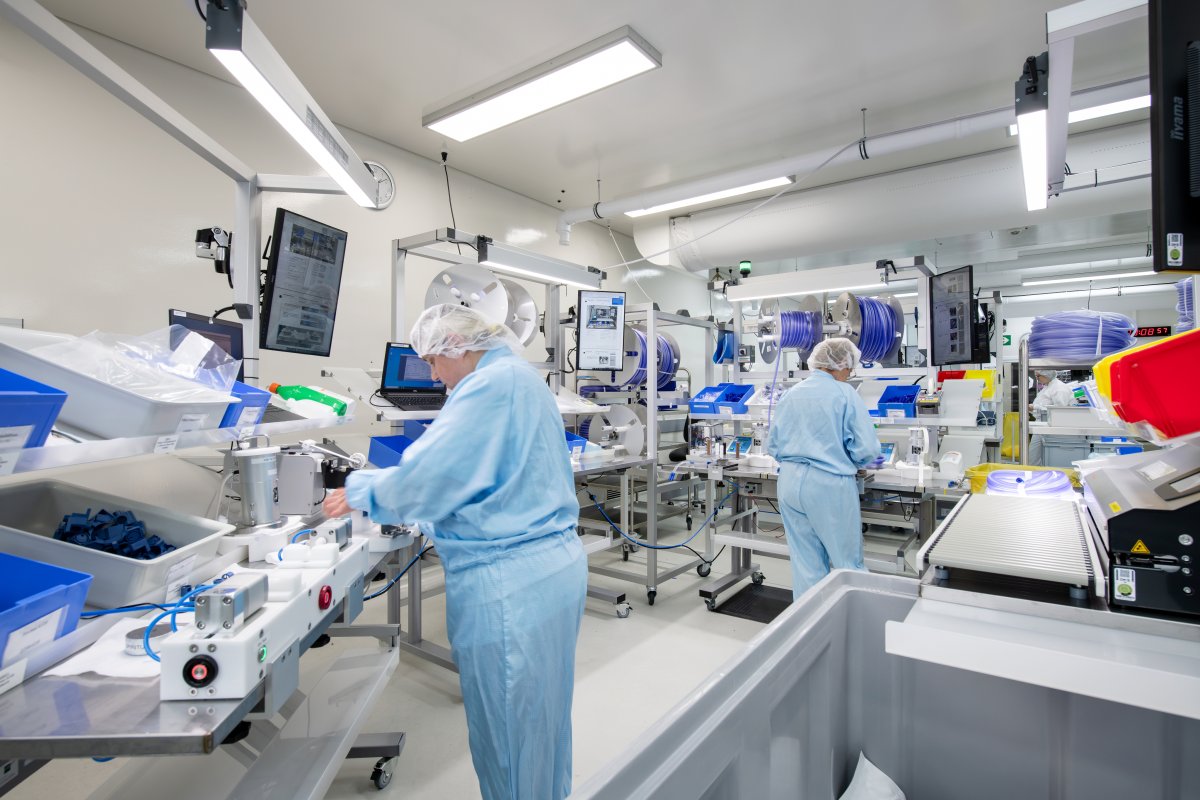
Precision begins at the Core
Every life-saving procedure starts with uncompromising quality.
Our components are engineered with precision, using advanced materials and rigorous testing to ensure flawless performance in the most critical environments.
Support that
never Sleeps.
Our commitment doesn’t end in the OR.
With proactive maintenance, rapid response teams, and global service coverage, we ensure your devices perform flawlessly—day after day, procedure after procedure.
Crafted for Confidence
Expert hands and automated precision come together in our assembly process.
Each device is built to exacting standards, ensuring seamless integration and reliability when it matters most—inside the operating room.
Protected.
Delivered.
Ready.
From sterile barrier systems to shock-resistant logistics:
Our packaging and shipping protocols guarantee that every device arrives hospital-ready-safe, secure, and compliant with global medical standards.
Empowering Surgeons. Elevating Outcomes.
In the hands of skilled professionals, our devices enable minimally invasive procedures that reduce recovery time and improve patient outcomes.
Trusted by hospitals worldwide, we’re proud to be part of every successful intervention.
Handled by us worldwide
As a leader in Minimally Invasive Medicine, we are familiar with the high safety demands that are placed on products worldwide. This is especially true with approvals and registrations by national authorities. As part of our services, we support you with our experience and expertise across borders.

CE, FDA, CFDA – all of the important Seals of Approval
We manage global approval processes for you—covering CE marking in Europe, FDA registration in the U.S., and certifications in Asia, including CFDA and PMDA and more.
With 40 FDA approvals to date, our track record speaks for itself. Trust Novanta to navigate every regulatory step with expertise.

Approvals and Registrations of Medical Products
As a leader in Minimally Invasive Medicine, we are familiar with the high safety demands that are placed on products worldwide. This is especially true with approvals and registrations by national authorities.
As part of our services, we support you with our experience and expertise across borders.
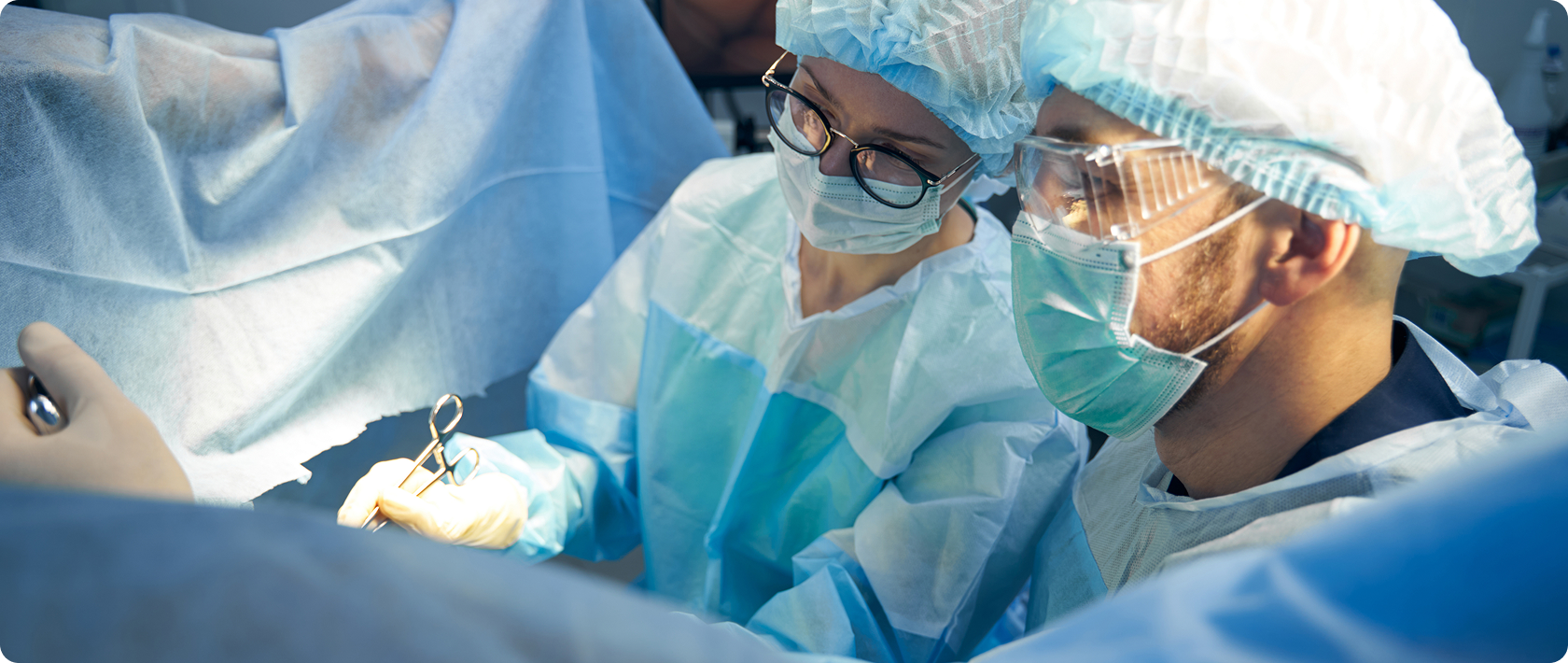
Protecting lives through precision Compliance
We build trust through rigorous quality systems, ensuring every product meets global regulatory standards and delivers safe, effective solutions that healthcare professionals and patients rely on.
Our other products

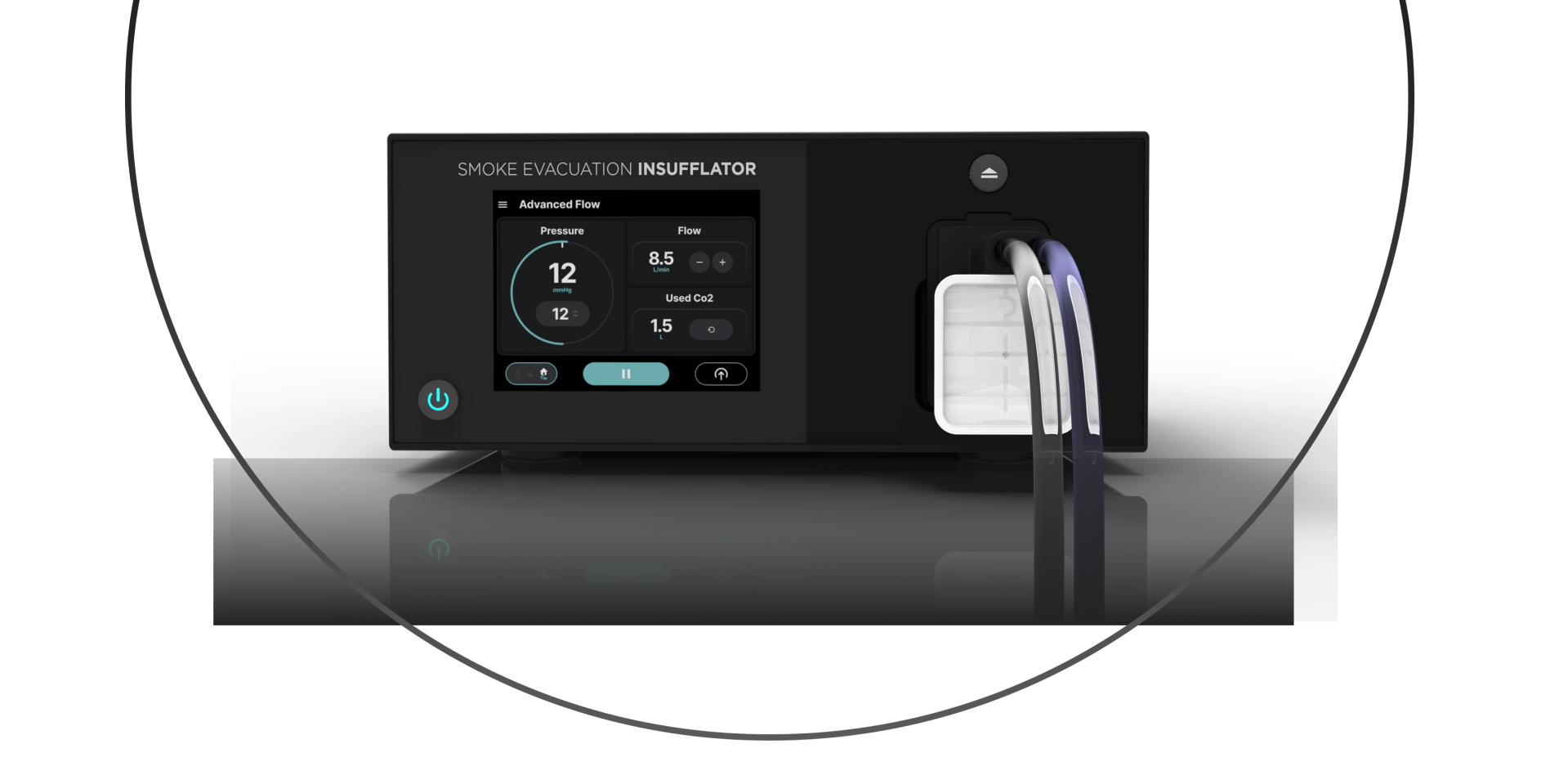
Advanced Surgery
Insufflators

Advanced Surgery
Pumps

Advanced Surgery
iOR

Advanced Surgery